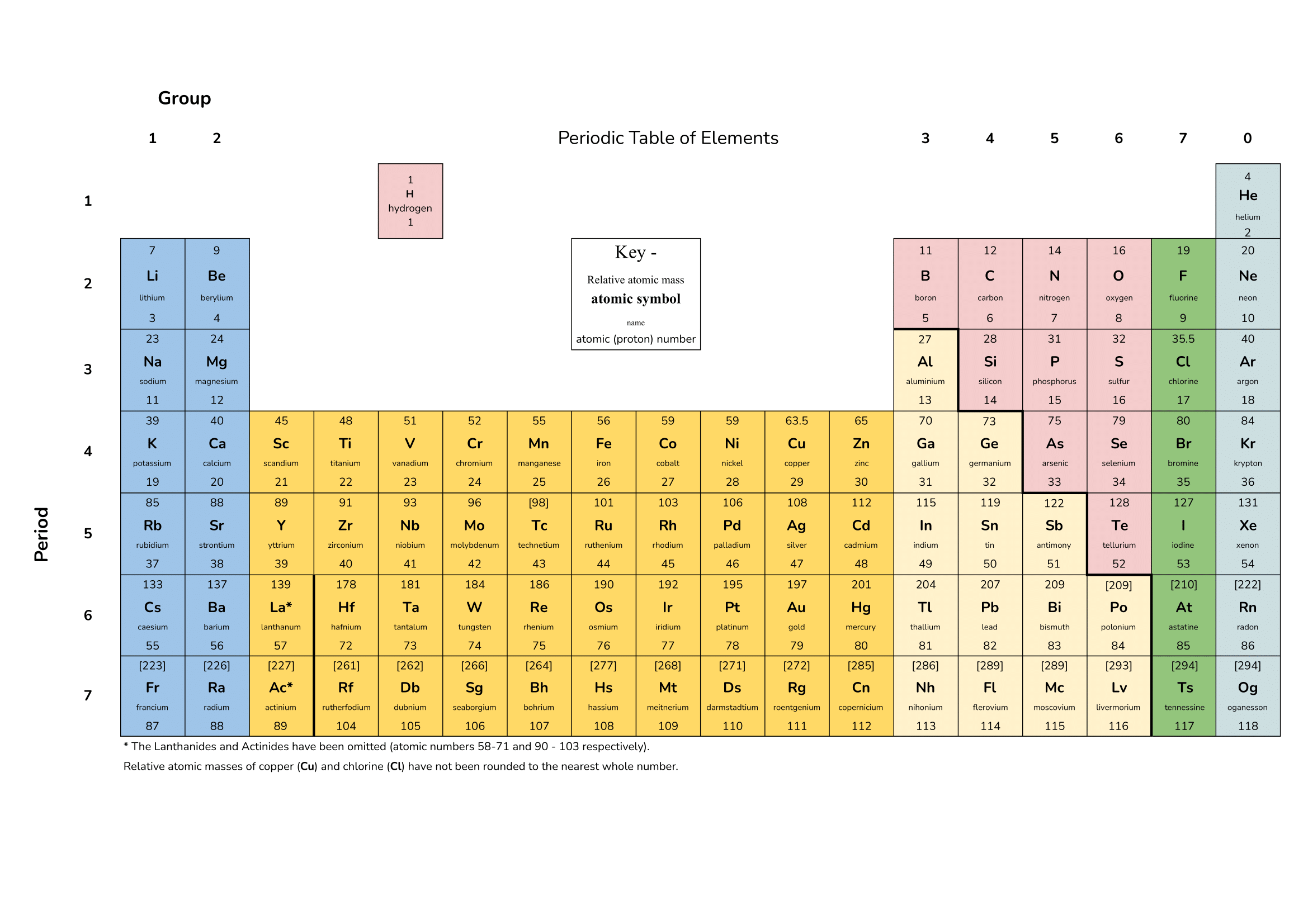
Do you know what Mᵣ stands for?
Relative Atomic Mass and Relative Formula Mass
How to Calculate Relative Atomic Mass
1) Find the abundance (percentage) of each isotope: Cl-35 (75%) and Cl-37 (25%)
2) Multiply mass number by percentage for each and sum together: 35 x 75 + 37 x 25
3) Divide the total by 100 to find the relative atomic mass (Ar): 3550 / 100 = 35.5
What is Relative Atomic Mass?
Relative atomic mass (Aᵣ) is the mean mass of all of the isotopes of an element. There are many different isotopes of each element and the masses of those which are stable (do not decay over time) can be averaged to find this number.
To calculate Aᵣ you should multiply the mass of each isotope by its relative abundance and add the results together.
Example Aᵣ calculation:
Lithium has two stable isotopes, Li-6 (abundance – 1.9%) and Li-7 (abundance – 98.1%).
(6 x 0.019) + (7 x 0.981) = Aᵣ 6.981
What is Relative Formula Mass?
Relative formula mass (Mᵣ) is the sum of the relative atomic masses (Aᵣ) shown in the molecular formula.
Mᵣ is used to determine multiple other important values in chemistry, such as the empirical formula if given the masses of the elements.
Example Mᵣ calculation:
Question: Find the relative formula mass of CO₂.
Add together the Aᵣ of each element, multiplied by the number of its atoms in the compound:
Carbon Aᵣ = 12. Oxygen Aᵣ = 16
12 + (2 x 16) = Mᵣ 44
For more practice Mᵣ and Aᵣ calculations visit this page from the Oregon Institute of Technology
We hope you have enjoyed this quiz on relative atomic and relative formula masses.
For more fundamental chemistry knowledge, check out the atomic structure quiz and the elements, compounds and mixtures quiz.