Do you know the difference between an element, a compound and a mixture?
Elements, Compounds and Mixtures Quiz
Quiz Summary
0 of 8 Questions completed
Questions:
Information
You have already completed the quiz before. Hence you can not start it again.
Quiz is loading…
You must sign in or sign up to start the quiz.
You must first complete the following:
Results
Results
0 of 8 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 8
1. Question
Which of the following describe the composition of a pure substance?
CorrectIncorrect -
Question 2 of 8
2. Question
Which types of atoms can be found in a pure piece of gold?
CorrectIncorrect -
Question 3 of 8
3. Question
[question number = 01.5] Drag and drop to match the description to the term
Sort elements
- Element
- Compound
- Mixture
- Contains only a particular type of atoms
- Contains more than one type of atoms, chemically bonded together
- Contains more than one type of atoms, but not chemically bonded together
CorrectIncorrect -
Question 4 of 8
4. Question
Drag and drop to match the substance to the correct type
Sort elements
- Mixture
- Compound
- Element
- Air
- Carbon dioxide
- Pure gold
CorrectIncorrect -
Question 5 of 8
5. Question
Use one of the following words to fill in the blank:
Solvent, solute, solution-
A is a mixture made of dissolved substances in a liquid.
CorrectIncorrectHint
Use one of the following words:
solution
solute
solvent
solent
-
-
Question 6 of 8
6. Question

The graph shows a heating curve for substance X (red) and substance Y (blue). Drag and drop to match the substance to the correct description.
Sort elements
- X (red)
- Y (blue)
- Pure
- Impure
CorrectIncorrect -
Question 7 of 8
7. Question

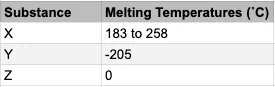
The table shows 3 substances and their melting points. Drag and drop to match the substance to the correct type.
Sort elements
- Mixture
- Element or compound
- X
- Y
CorrectIncorrect -
Question 8 of 8
8. Question

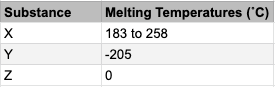
The table shows 3 substances and their melting points. What common substance, in particular, could substance Z be?
CorrectIncorrectHint
Which common, very important substance melts or freezes at 0 degrees C?
Read our post on elements, compounds and mixtures to learn about their differences and why they are important in chemistry.
Which exam board are you studying?
Loading...
Submit Your Answer
Scan with your phone: