What do you know about alkanes?
Alkanes and Cracking Quiz
Good Luck!
Quiz Summary
0 of 9 Questions completed
Questions:
Information
You have already completed this quiz. You cannot start it again.
Quiz is loading…
You must sign in or sign up to take this quiz.
You must first complete the following:
Results
Quiz complete. Results are being recorded.
Results
0 of 9 Questions answered correctly
Your Time:
Time has elapsed.
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average Score |
|
| Your Score |
|
Categories
- Not categorized 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 9Question 1
Alkanes and alkenes are both part of a series, what is the name we give to the series?
CorrectIncorrect -
Question 2 of 9Question 2
Alkanes all have the same general formula. What is that formula?
CorrectIncorrect -
Question 3 of 9Question 3
What is the formula that represents how much each alkane differs from each neighbouring compound?
CorrectIncorrect -
Question 4 of 9Question 4
Select the accurate statements about alkanes.
CorrectIncorrect -
Question 5 of 9Question 5
Which of the following is a difference between alkanes and alkenes?
CorrectIncorrect -
Question 6 of 9Question 6
What is the purpose of cracking?
CorrectIncorrect -
Question 7 of 9Question 7
Crude oil separated by fractional distillation creates unequal volumes of each fraction e.g. fuel oil. These unequal volumes also do not match the demand for each fraction. What process is carried out to balance out the supply with demand?
CorrectIncorrect -
Question 8 of 9Question 8
Which descriptions are accurate about saturated and unsaturated hydrocarbons?
CorrectIncorrect -
Question 9 of 9Question 9
The following equations show cracking of a long hydrocarbon into a shorter alkane and an alkene. Which equation is correct?
CorrectIncorrect
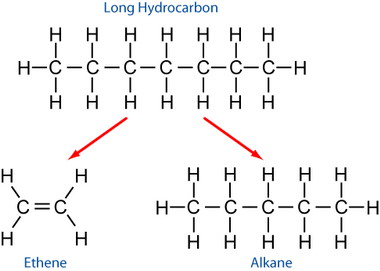
How does cracking work?
Cracking breaks down large alkanes, meaning alkanes with very long carbon chains, into smaller alkanes as well as an alkene. Look at the example to understand how this happens. Notice that the equation for this reaction would be balanced since there is the same number of atoms of products as reactants.
Which exam board are you studying?
