What is the Avagadro Constant?
Moles Quiz
What are Moles?
Moles are the SI unit for the amount of a substance.
The symbol for moles is mol.
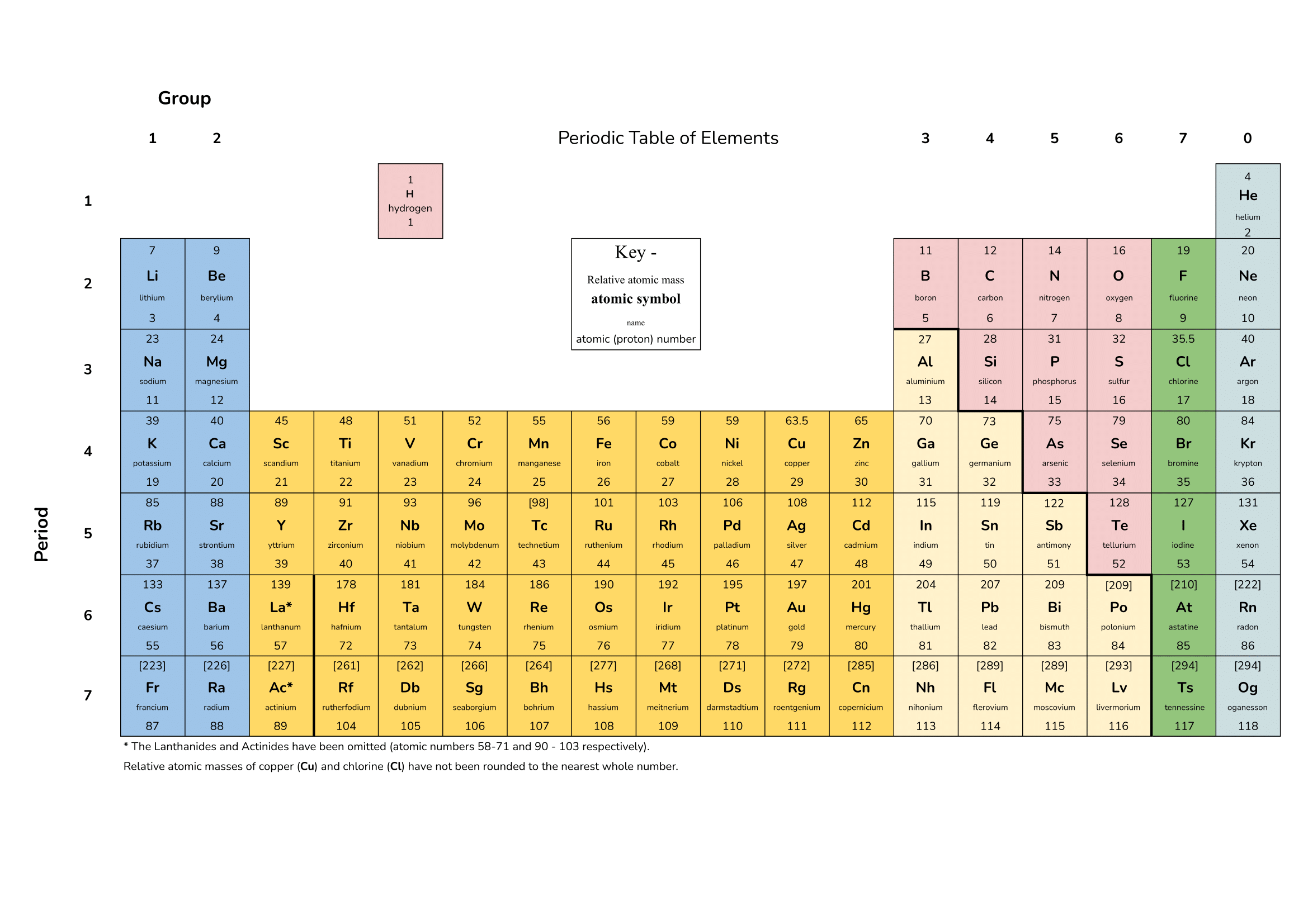
There are the Avagadro’s Constant number of particles in a mole of any substance. This can be atoms, ions or molecules. The Avagadro’s Constant is 6.02 x 10²³. So there are that many atoms in a mole of carbon or the same number of molecules in a mole of water.
You can calculate the number of moles using the following equation:
number of moles = mass of substance (g) / Aᵣ or Mᵣ
Rearrange this equation to get the mass or A/Mᵣ given the other two numbers.
How to determine a balanced chemical equation from the masses of reactants and products.
To do this you should –
- Find out the Mᵣ or Aᵣ of each reactant and product.
- Determine the number of moles of each reactant and product, do this using the equation in the section above “What are moles?”.
- Divide the number of moles of each reactant and product by the smallest number of moles.
- Multiply each number to make sure you have a whole number – the simplest whole number ratio.
- Write the equation as normal
- Balance the equation
Example: Determine the balanced symbol equation for a reaction between 80g of sodium hydroxide (NaOH) and 73g of hydrochloric acid (HCl) to produce 117g of sodium chloride (NaCl) and water (H₂O).
- Mᵣ of NaOH = 40, Mᵣ of HCl = 36.5, Mᵣ of NaCl = 58.5
- NaOH = 2 mol, HCl = 2 mol, NaCl = 2 mol
- The smallest number of moles = 2, so divide each by 2
- 1 mol of NaOH and 1 mol of NaCl makes 1 mol of NaCl
- NaOH + HCl → NaCl + H₂O
Since we don’t usually put 1 before chemical symbols in an equation, it is already balanced.
This is a relatively simple example where all of the substances were in the same number of moles and no work is needed to balance the equation. Follow the same procedure for more complex examples and you will be able to do these problems.
You can always search on YouTube for more examples by entering:
“Using moles to balance equations”